health
February 21, 2026
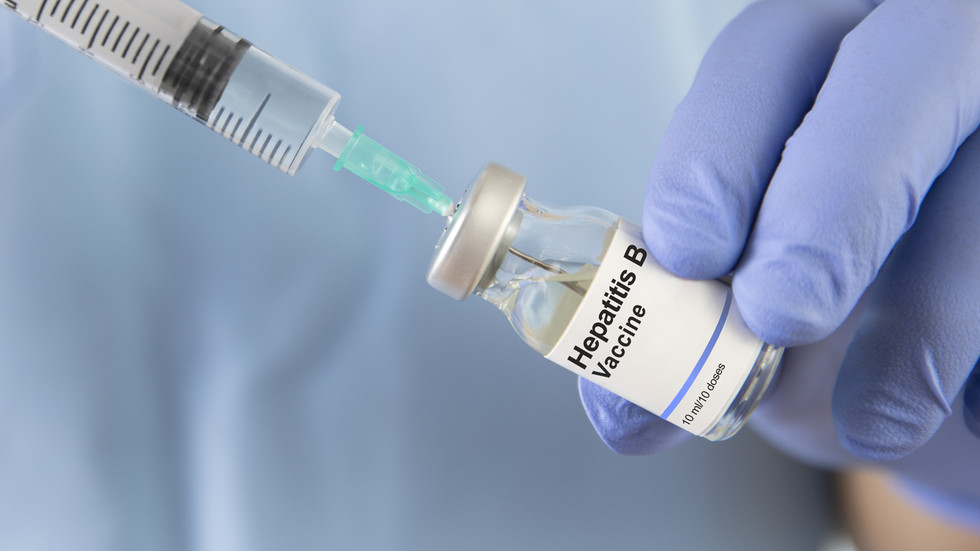
African state cancels US-funded vaccine study
Guinea-Bissau’s foreign minister has announced that a US-backed hepatitis B trial will not go ahead due to ethical concerns
TL;DR
- Guinea-Bissau has stopped a controversial hepatitis B vaccine study funded by the US.
- The World Health Organization (WHO) raised ethical concerns about the trial's design.
- The study involved withholding the hepatitis B vaccine from half of the newborns at birth.
- The WHO denounced the trial as "unethical" and warned of potential harm.
- The study was backed by a $1.6 million grant from the US Centers for Disease Control and Prevention.
Continue reading the original article